Peripheral ulcerative keratitis in Sweet syndrome
Denise Ramona Carbonell, MD, Maria Cecilia Gertrudis Agdeppa, MD, DPBO, Margarita Nahleen N. Mejia, MD
SWEET syndrome (acute febrile neutrophilic dermatosis) is a skin disorder characterized by bluish-red papules or cutaneous red nodules accompanied by circulating neutrophilia, and an infiltrate of mature neutrophils in the upper dermis. It is typically a dermal variant of neutrophilic dermatosis, the features of which were first described in 5 patients by Dr. Robert Sweet in 1964. Although Sweet syndrome is primarily a skin disorder, sterile neutrophilic infiltrates may also localize in other organs, such as bone, lung, liver, spleen, brain, and eye. Ocular inflammation, including peripheral ulcerative keratitis, is reported to occur in about one-third of patients but few cases have been described in detail. We reviewed a case of a 31-year-old male with Sweet syndrome presenting with peripheral ulcerative keratitis (PUK).
CASE REPORT
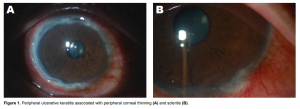
A 31-year-old Micronesian male, diagnosed with Sweet syndrome in late 2009, was on unsupervised steroid treatment for months. He was admitted for pulmonary nodules secondary to granulomatous tuberculous infection and treated with anti-TB drugs. He also had increased skin lesions, noted on upper and lower extremities and abdomen, considered as fungal pyoderma. He was started on antifungal medications. A month after, the patient was referred to Ophthalmology for a painful, red, right eye (OD), that started as a nodular perilimbal hyperemic to purplish lesion at the 6 o’clock position, associated with increased photosensitivity and severe, boring, right-sided ocular pain. Presence of a rheumatologic problem was ruled out by laboratory exams. Clinical impression was nodular scleritis OD. He was treated with piroxicam 20 mg twice a day, moxifloxacin (Vigamox) and nepafenac (Nevanac) 4 times a day in OD. There was increased ocular pain despite these medications with development of new nodular perilimbal lesions, more hyperemic at the edges at 12 and 3 o’clock positions. Nepafenac was discontinued and cyclosporine (Restasis) and carboxymethylcellulose (CMC) preservative- free (Cellufresh) every 2 hours were started. As his pulmonary TB and fungal infections were responding to treatment, IV methylprednisolone 1gram/day for 3 days was initiated. It was discontinued after the third day due to worsening of the cutaneous lesions. Patient was discharged with previous management and instructed to follow-up closely. A month later, there was gradual peripheral corneal thinning starting at the 7 to 9 o’clock region of the right eye and spreading circumferentially (Figure 1A). There was also beginning scleritis, seen as deep hyperemia at the 5 o’clock region, of the left eye (OS) (Figure 1B). Vision remained at 20/20 both eyes (OU). Clinical impression was peripheral ulcerative keratitis (PUK) associated with scleritis OU. Patient was readmitted for pulsed IV methylprednisolone 1 gram/day for 3 days; moxifloxacin, cyclosporine and CMC were applied every 2 hours OU. Piroxicam was discontinued. A soft bandage contact lens was applied in OD as a prophylactic measure against an inadvertent perforation from the peripheral corneal thinning. Patient was discharged after the third IV methylprednisolone and started on oral prednisone 60mg/day. On follow-up, there was gradual vascularization and healing of the lesion in OS. In OD, the peripheral corneal thinning was spreading centripetally (Figure 2A and B). Vision decreased to 20/400 OD, 20/20 OS. Oral prednisone was tapered and oral cyclosporine at 2.5mg/day was started. All eye medications were continued. Anterior segment photographs were taken. After one month, pannus formation with fibrosis and opacification of the peripheral cornea was seen in OD (Figure 3A). The vision improved to 20/25 withsignificant astigmatism (plano-2.25 cyl x 12). There was complete resolution of the PUK in OS (Figure 3B). Intraocular pressures were normal for both eyes. Oral cyclosporine was discontinued after the white-cell count decreased. Prednisone was maintained at 20mg/day for persistent skin lesions. Patient was advised frequent follow-ups to monitor ocular effects of prolonged corticosteroid use.
DISCUSSION
Sweet syndrome presents in three clinical settings.1 The diagnosis is based on the presence of typical skin findings with histopathological confirmation of a cutaneous neutrophilic infiltrate. The plaques are usually painful at onset and typically spread from the head and neck area to the face, trunk, and limbs. Isolated lesions may be seen on the legs, wrists, hands, or fingers.2 Along with symptoms of malaise, fatigue, and fever, seronegative inflammatory arthritis is a common feature. The presentation of Sweet syndrome in conjunction with inflammatory disease is of particular concern to ophthalmologists. Inflammatory conditions can be found in about 15% of patients. Crohn’s disease and ulcerative colitis, which some authors consider part of a spectrum of neutrophilic disease, are the most common.3 Sjogren syndrome, Behcet disease, systemic lupus erythematosus, and rheumatoid arthritis may also be associated. About 15 to 20% of cases can occur in the setting of malignancy, particularly hematologic malignancies such as acute myeloid leukemia (AML) and monoclonal gammopathies.4 Ocular inflammation is said to occur in about onethird of patients with Sweet syndrome.3 Conjunctivitis and other types of inflammation, including iritis, episcleritis, and peripheral ulcerative keratitis, demonstrate the range of ocular involvement in Sweet syndrome. Few reports were mentioned pertaining to these three sites of ocular inflammation. Our patient presented with nodular scleritis and peripheral ulcerative keratitis throughout the course of his disease. Peripheral ulcerative keratitis (PUK) is a potentially destructive disorder comprising of a crescent-shaped destructive inflammation at the margin of the corneal
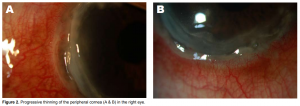
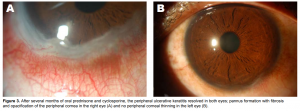
Figure 2. Progressive thinning of the peripheral cornea (A & B) in the right eye. Figure 3. After several months
of oral prednisone and cyclosporine, the peripheral ulcerative keratitis resolved in both eyes; pannus formation with fibrosis and opacification of the peripheral cornea in the right eye (A) and no peripheral corneal thinning in the left eye (B). stroma, associated with epithelial defects, presence of stromal inflammatory cells, and progressive stromal degradation and thinning. PUK can rapidly produce progressive corneal stromal necrosis, leading to perforation and eventual blindness. The peripheral cornea has distinctive morphologic and immunologic features making it susceptible to inflammatory reactions. Compared to the avascular central cornea, the peripheral cornea is closer to limbal conjunctiva and receives part of its nutrient supply from limbal capillary arcade, which is a source of immunocompetent cells. Any inflammatory stimulus in the peripheral cornea, in this case the cutaneous neutrophilic infiltrates, may produce local and systemic immune responses resulting in neutrophil recruitment and complement activation of both classic and alternative pathways in tissue and vessel. Activated complement components increase vascular permeability and produce chemotactic factors for neutrophils. These neutrophils infiltrate the peripheral cornea and release proteolytic and collagenolytic enzymes, reactive oxygen metabolites, and proinflammatory substances, causing degradation of the corneal stroma. Inflamed limbal conjunctiva is also capable of producing collagenase, contributing to stromal degradation. The recommended treatment for PUK includes ruling out secondary infection, treating existing keratitis sicca, and systemic immunosuppression.
The acute inflammation is best managed with oral or intravenous corticosteroids with simultaneous initiation of a maintenance immunosuppressive agent, such as cyclosporine or methotrexate. The use of systemic treatment in this patient is critical for the resolution of the ocular inflammation, as well as its systemic involvement.5 As in other cases reported, our patient’s ocular manifestations improved with systemic administration of corticosteroids. In most cases of Sweet syndrome, prednisone is extremely and rapidly effective, in doses of 1 mg/kg/d. Despite initial favorable response, recurrences of neutrophilic dermatitis are common and generally develop as steroid use is being tapered. Once the underlying disease flares up, tapering of corticosteroid may take longer. Recurrence rates of 25 to 30% have been reported.1-2 Our patient initially responded well with intravenous corticosteroids but cutaneous skin lesions flared progressively and fungal elements started to develop, halting treatment. High-potency topical steroids or intralesional glucocorticoids may also be useful in localized lesions.2 Wilson and colleagues reported a patient with peripheral ulcerative keratitis and episcleritis successfully treated with oral cyclosporine. Oral prednisone or prednisolone are the mainstays of treatment for Sweet syndrome. It appears that topical corticosteroids are not required if systemic treatment is initiated.6 Steroid-sparing agents may be helpful in the long run to prevent unwanted complications. Many medications act on inhibiting neutrophil chemotaxis, but none has been shown to be as effective as corticosteroids. The use of topical nonsteroidal antiinflammatory agents, however, has been questionable. Some reports cited that they were not as effective as immunosuppresants like cyclosporine.5,6 Patients may benefit from immunosuppressive agents. In the study of Kaçmaz and Kempen, most patients tolerated cyclosporine and toxicity was more frequent in those 55 years and above. Cyclosporine had a high rate of side effects with nephrotoxicity in early trials using higher dosage. Currently, the recommended dosage is 3 mg/ kg/day or less.7 In summary, Sweet syndrome has been reported in association with many diseases, including ocular inflammation. Peripheral ulcerative keratitis is a potentially blinding disorder that can be seen in Sweet syndrome. With good clinical history, meticulous ocular examination, initiating treatment with corticosteroids and other immunosupressive agents, and close monitoring through frequent follow-ups, PUK can be treated and cured.
1.Cohen PR, Talpaz M, Kurzrock R. Malignancy-associated Sweet’s syndrome: review of the world literature. J Clin Oncol 1988; 6: 1887-1897.
2.Kato T, Kunikata N, Taira H, et al. Acute febrile neutrophilic dermatosis (Sweet’s syndrome) with nodular episcleritis and polyneuropathy. Int J Dermatol 2002; 41:
3.Von den Driesch P. Sweet’s syndrome (acute febrile neutrophilic dermatosis). J
4.Kemmet D, Hunter JA. Sweet’s syndrome: a clinicopathologic review of twentynine cases. J Am Acad Dermatol 1990; 23: 503-507.
5.Gottsh J, Apek E. Topical cyclosporine stimulates neovascularization in resolving sterile rheumatoid central corneal ulcers. Trans Am Ophthalmol Soc 2000; 98:
6.Wilson DM, John GR, Callen JP. Peripheral ulcerative keratitis—an extracutaneous neutrophilic disorder: report of a patient with rheumatoid arthritis, pustular vasculitis, pyoderma gangrenosum, and Sweet’s syndrome with an excellent response to cyclosporine therapy. J Am Acad Dermatol 1999; 40: 331-334.
7.Kaçmaz R, Kempen J, Cyclosporine for ocular inflammatory diseases. Ophthalmology 2010; 117: 576-584.

