Bonding strengths of 2-octyl cyanoacrylate, N-butyl-2- cyanoacrylate, and nylon 10-0
Jocelyn R. Elises, MD, Leo DP Cubillan, MD
LACERATIONS and perforations of the cornea are considered ophthalmic emergencies. Trauma is the leading cause of partial- and full-thickness corneal lacerations. Conditions such as microbial keratitis, keratoconjunctivitis sicca, neurotrophic or exposure keratopathy, and corneal ectasias may lead to corneal perforations. Disastrous sequelae of corneal lacerations and perforations include corneal scarring, infection, synechiae formation, cataract, glaucoma, and blindness. Prompt and effective treatment of these lacerations and perforations lessens the risk of these complications. Treatment options for corneal lacerations and perforations include suturing, corneal patch grafting, penetrating keratoplasty, keratoprosthesis, and closure
with tissue adhesives.1 In corneal lacerations, particularly in perforations, the maintenance of a watertight anterior chamber with no synechiae is important. Perforations are sealed with agents that appose wound edges and keep them watertight. The method mostly used to seal such perforations is suturing.2 The use of sutures in sealing corneal perforations is, however, not without complications. In sealing corneal lacerations, suturing has been known to cause significant astigmatic error as well as cheesewiring, inflammation, loosening, neovascularization, necrosis, and infection. When such complications arise, the patient will have to undergo suture removal, which in children and uncooperative adults is difficult to do.2 Sutureless procedures using glues are advantageous because these techniques immediately restore the integrity of the globe and decrease the risk of complications. Biologic glues include fibrin tissue adhesive or fibrin glue (Beriplast P, Aventis) while N-Butyl-2-cyanoacrylate (Histoacryl, Braun) is the more commonly used chemical glue for corneal surgery (off-label use). Fibrin glue and N-butyl-2-cyanoacrylate as tissue adhesives for corneal perforations have been found effective in closing corneal perforations up to 3 mm, although fibrin glue takes a longer time for adhesive plug formation than N-butyl-2 cyanoacrylate.3 Commercially produced fibrin tissue adhesive from pooled blood is biodegradable, but because of fear of viral contamination with hepatitis B, hepatitis C, or the human immunodeficiency virus, it is not approved by the United States Food and Drug Administration (FDA).4 Cyanoacrylate adhesives were first described in 1949. Cyanoacrylate forms a strong bond once it polymerizes. Polymerization of the adhesive occurs when water or any basic substance gets in contact with it, producing an exothermic reaction. Over the years, several cyanoacrylate monomers have been developed as tissue adhesives. However, their clinical use has been limited because of their physical properties.
Butyl cyanoacrylate derivatives are commercially available tissue adhesives not yet approved by the FDA for clinical use. Cyanoacrylate tissue adhesives have many favorable properties—including strong bonding, short polymerization time, and relatively low cost—that some ophthalmologists use it for ocular surgery. Unfortunately, some cyanoacrylate derivatives can cause histotoxic effects because they degrade into formaldehyde and cyanoacetate. The degree of acute inflammation is inversely proportional to the length of the monomer chain. Among the longer-chain derivatives is 2-Octyl cyanoacrylate, a combination of monomers and plasticizers that forms a strong flexible bond. It has a three-dimensional breaking strength four times that of N-butyl-2-cyanoacrylate. 2-Octyl cyanoacrylate shows lower toxicity than N-butyl-2- cyanoacrylate because of decreased by-product formation of formaldehyde.5, 6, 7 In 2001, the FDA approved the use of 2-Octyl cyanoacrylate in sealing small skin incisions and lacerations. It naturally sloughs off the skin in 5 to 10 days. It is also 99-percent effective in protecting against Staphylococcus epidermidis, Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa and Enterococcus faecium, acting as a microbial barrier. After the wound has been cleaned, the edges are apposed and 2-Octyl cyanoacrylate is applied in layers (minimum of 3) with a light brushing stroke. It forms a strong, flexible bond in just 45 to 60 seconds and reaches full strength in approximately 2 minutes and 30 seconds. The incision site need not be kept dry.8 Successful treatment of a corneal perforation with 2-Octyl cyanoacrylate has been reported by Taravella et al. After application of the glue, the anterior chamber deepened and the visual acuity improved. The glue remained intact for 6 weeks and eventually fell off.9 Since 2-Octyl cyanoacrylate forms a waterproof dressing, it may even be used on frequently wet surfaces like the cornea. To understand the sealing properties of cyanoacrylate adhesive, we compared the bonding strengths of corneal perforations sealed with 2-Octyl cyanoacrylate (Dermabond, Johnson & Johnson, New Brunswick, NJ, USA), N-Butyl-2-Cyanoacrylate (Histoacryl, B. Braun, Melsungen, Germany), a commonly used adhesive, and Nylon 10-0 (Alcon, Fort Worth, TX, USA), the gold standard, by determining the leaking pressures as measured by a manometer attached to the anterior-chamber maintainer of the eye.
METHODOLOGY
This is a single blind, completely randomized physical experimental animal study. The sample size per treatment group of 13 eyes was computed using a power of 95%, confidence interval of 5%, standard deviation of 6, and a maximum tolerable level of 7 mm Hg. Seventy-eight freshly enucleated porcine eyes with average corneal diameter of 14.5 mm and baseline Schiotz
intraocular pressure of 17.3 mm Hg were randomly assigned to the 3.0 mm (n=39) and the 5.1 mm perforation groups (n=39). All perforations were made perpendicular to the corneal surface with a fresh 3.0 mm or 5.1 mm metal keratome at the central portion of the cornea. Thirteen corneal perforations in each group were sealed either with nylon 10-0 sutures, Dermabond, or
Histoacryl by random allocation. In the sutured corneas, the 3.0 mm perforations were sealed with 1 suture while the 5.1 mm perforations were sealed with 2 sutures. In
those sealed with Dermabond, the wound edges were dried and the tissue adhesive was applied thinly 3 times using the applicator tip with a brushing motion parallel to the corneal perforation. In those sealed using Histoacryl, the wound edges were dried and a drop of tissue adhesive was applied and spread across the perforation. In both Dermabond and Histoacryl groups, the eye was irrigated with normal saline solution after application of the tissue adhesive for polymerization to occur. An anterior chamber maintainer was inserted through a 1 mm stab incision. Intraocular pressure was raised by injecting normal saline through the anterior-chamber maintainer immediately after suturing and 3 minutes after polymerization of both Dermabond and Histoacryl. A manometer, precalibrated up to 100 mm Hg, was attached to the end of the anterior-chamber maintainer to determine the intraocular pressure. The post-sealing intraocular leaking pressures were recorded with the reader blinded as to the treatment groups. Wound leak was determined using Seidel’s test. No leak was recorded if the Seidel’s test was negative when the intraocular pressure was raised to 100 mm Hg, the highest reading in the precalibrated manometer used. The proportion of eyes that leaked and the mean leaking pressures were computed. Fisher’s Exact Test (Stata Corporation, College Station, TX, USA) was used to determine the difference in the proportion of eyes that leaked between the treatment groups. Wilcoxon signed ranked test was used to compare the mean leaking pressures of eyes that leaked between the treatment groups.
RESULTS
Included were 78 porcine eyes, distributed equally into 3 mm and 5.1 mm perforation groups. Each perforation group was divided into three types of sealing methods: 2-Octyl cyanoacrylate, N-butyl-2-cyanoacrylate, and nylon 10-0; total of 13 samples each. In the 3 mm corneal perforation group, only 2 (15.4%) of the 13 eyes sealed with 2-Octyl cyanoacrylate leaked (Table 1). Mean leaking pressure was 79.5 mm Hg. Only 1 (7.7%) of the 13 eyes sealed with N-butyl-2-cyanoacrylate leaked. Leaking pressure was 88.0 mm Hg. All of the 13 eyes sutured with nylon 10-0 leaked. Mean leaking pressure was 61.44 mm Hg. The proportions of eyes that leaked in both the 2-Octyl cyanoacrylate and the N-butyl-2- cyanoacrylate groups were comparable (p = 1.00). The proportion of eyes that leaked in the nylon group, however, was significantly higher (p < 0.001) than the other 2 groups. The mean leaking pressure of 2-Octyl cyanoacrylate was higher compared with that of the nylon 10-0 group (p = 0.16). Similarly, the leaking pressure of the N-butyl-2-cyanoacrylate group was higher than the nylon 10-0 group (p = 0.32). In the 5.1 mm corneal perforation group, 4 (30.8%) of the 13 eyes sealed with Dermabond leaked (Table 1). Mean leaking pressure was 92.75 mm Hg. In the Histoacryl group, 2 (15.4%) of the 13 eyes leaked. Mean leaking pressure was 86.5 mm Hg. All of the 13 eyes sutured with nylon 10-0 leaked. Mean leaking pressure was 59.08 mm Hg. The proportions of eyes that leaked in both the Dermabond and Histoacryl groups were comparable (p = 0.65). The proportion of eyes that leaked in the nylon 10-0 group, however, was significantly higher (p < 0.001) compared with both the Dermabond and Histoacryl groups. The mean leaking pressure of Dermabond was higher compared with the nylon 10-0 group (p = 0.07). Similarly, the mean leaking pressure of the Histoacryl group was higher compared with the nylon 10-0 group (p = 0.10).
DISCUSSION
Bonding strength is the ability of the tissue edges to remain apposed and maintain a watertight anterior chamber in corneal perforations. In this study, bonding strength was determined by measuring the leaking pressures of sealed experimental perforations. Bonding strengths of nylon 10-0, Dermabond, and Histoacryl were determined and compared. Results showed that in the 3.0 mm perforation group, the mean leaking pressure of Dermabond was lower than
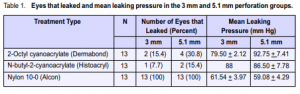
that of Histoacryl but higher than that of nylon 10-0. In the 5.1 mm perforation group, however, the mean leaking pressure of Dermabond was higher compared with those of Histoacryl and nylon 10-0. The differences in mean leaking pressures, however, were not statistically significant. The results of this study do not support the findings by Quinn in 1997 that compared sutures and 2-Octyl cyanoacrylate in sealing skin lacerations. Quinn reported that N-butyl-2-cyanoacrylate formed a strong bond that was brittle and prone to fracture when used on long lacerations. 2-Octyl cyanoacrylate has a longerchain formulated cyanoacrylate containing monomer and plasticizers. This produces a stronger, flexible, 3-dimensional bond more suited for longer lacerations. The study concluded that 2-Octyl cyanoacrylate is more effective than N-butyl-2-cyanoacrylate for longer skin lacerations.5 Our study showed that both have comparable bonding strengths and are effective in sealing 3 mm and 5.1 mm corneal perforations. The proportion of leak among porcine eyes sealed with Dermabond is significantly lower compared to that of eyes sealed with nylon 10-0. The proportion of leak among porcine eyes sealed with Dermabond is comparable to that of Histoacryl. This difference is not statistically significant. In this study, the bonding strengths (in terms of leaking pressure) of corneal perforations sealed with 2-Octyl cyanoacrylate and N-butyl-2-cyanoacrylate are comparable and greater than that of nylon 10-0. These two tissue adhesives are both effective in sealing 3.0 mm and 5.1 mm corneal perforations.
References
1. Miki D, Dastgheib K, Kim T, et al. A photopolymerized sealant for corneal lacerations. Cornea 2002; 21: 393-399
2. Santos M, Flores J. Wound strength of experimental corneal perforations sealed with fibrin glue in cadaver porcine eyes. Philipp J Ophthalmol 2002; 27: 59-63.
3. Sharma A, Kaur R, Kumar S, et al. Fibrin glue versus n-butyl-2-cyanoacrylate in corneal perforations. Ophthalmology 2003; 110: 291-298.
4. Siedentop K, Park J, Sanchez B. An autologous fibrin tissue adhesive with greater bonding power. Arch Otolaryngol Head Neck Surg 1995; 121:769-772.
5. Quinn J, Wells G, Sutcliffe T, et al. A randomized trial comparing octylcyanoacrylate tissue adhesive and sutures in the management of lacerations. JAMA 1997; 227: 1527-1530.
6. Macsai M. The management of corneal trauma: advances in the past twenty-five years. Cornea 2000; 19: 617-624.
7. Gosain AK, Lyon, VB. The current status of tissue glues: part II for adhesion of soft tissues. Plast and Reconstr Surg 2002; 110: 1581-1584.
8. Johnson & Johnson, Dermabond Topical Skin Adhesive. http://www.dermabond.com.htm (accessed July 30, 2003).
9. Taravella MJ, Chang CD. 2-Octyl cyanoacrylate medical adhesive in treatment of a corneal perforation. Cornea 2001; 20: 220-221.
Acknowledgment
The authors thank Drs. Amor Y. De la Cruz and Sheila Marie Lavina for their invaluable support.

