Long-Term Effect on Corneal Curvature of Corneal Collagen Cross-Linking for Keratoconus
Jess L. Paningasan, Jr., MD and Ruben Lim Bon Siong, MD
Eye Institute, St. Luke’s Medical Center
E. Rodriguez Ave., Quezon City, 1102, Philippines
Correspondence: Jess L. Paningasan Jr., MD
St. Luke’s Medical Center, E. Rodriguez Ave., Quezon City, 1102, Philippines
Email: j_paningasan@yahoo.com
Disclosure: The authors report no conflict of interests.
Keratoconus, a progressive corneal ectatic disorder, usually begins at puberty and stabilizes during the third to fourth decade of life.1 It presents clinically as progressive thinning of the corneal stroma producing a conical shape more pronounced at the central, paracentral, or inferotemporal portion of the cornea.1 The Collaborative Longitudinal Evaluation of Keratoconus study reported an increase of 1.6 diopter (D) over the flat meridian in 8 years. In the same time period, 24% of patients had at least 3.0 D increase in the steep meridian. Throughout the disease course, almost 20% of patients required keratoplasty.2
Treatment of keratoconus focuses on alleviating the visual impairment resulting from corneal thinning.3 Early in the disease process, contact lenses are the mainstay of therapy and treatment-of-choice over spectacles because they correct the irregular astigmatism induced by the cone-shaped cornea.3 However, more advanced disease necessitates surgical intervention which may include implantation of intrastromal corneal ring segments (ICRS), penetrating keratoplasty (PKP), and deep anterior lamellar keratoplasty (DALK).3 Keratoplasty only improves vision in keratoconus but does not treat the underlying cause. It is only by collagen cross-linking that progression of the disease can be halted.3
Collagen cross-linking is a process which occurs physiologically with age but can be photochemically induced with the use of riboflavin and ultraviolet A (UVA).4 Riboflavin acts as a photosensitizer by absorbing UVA and produces free radicals that induce collagen cross-linking through reactions with carbonyl groups specifically, the histidine, hydroxyproline, hydroxylysine, tyrosine, and threonine groups.4 However, the effects of collagen cross-linking are merely postulated because cross-links are not visualized under the microscope. Evidence that provide support to the effects of collagen cross-linking include increased stress strain measurements of corneal stromal tissue and increased resistance to matrix metalloproteinases following cross-linking.4
The first published study on the use of collagen cross-linking in the treatment of keratoconus was done in Dresden, Germany by Wollensak et al. in 2003. This study included 23 eyes of 22 patients with moderate or advanced progressive keratoconus. Study results showed regression in maximum K (Kmax) value and refractive error in 70% patients who underwent collagen cross-linking. Their study was the first to document disease regression. In the succeeding years, multiple, prospective, non-randomized studies further provided evidence of disease stabilization, significant post-operative improvements in visual acuity, and significant reductions in keratometry readings and higher-order aberrations following collagen cross- linking.4 Clinical studies supported the long-term efficacy of collagen cross-linking. However, there is no published local data regarding the effectiveness of collagen cross-linking for keratoconus. This study aimed to determine long-term changes on corneal astigmatism, best-corrected visual acuity (BCVA), and anterior and posterior elevation maps of keratoconus eyes that underwent corneal collagen cross-linking.
METHODS
This study was approved by the Institutional Scientific Review Committee. This was a descriptive, retrospective review of medical records of patients with keratoconus who underwent collagen cross-linking at St. Luke’s Medical Center – Quezon City and St. Luke’s Medical Center – Global City from January 2012 to March 2016. Demographic data, baseline and post-operative values of maximum keratometric value, BCVA, corneal astigmatism, and anterior and posterior elevation map changes were collected, analyzed, and compared.
Patients with pre-operative corneal topography consistent with keratoconus, clear corneas, keratometric value ≤60 D, and thinnest corneal thickness of at least 350 μm were included in the study. Patients included in the study also needed to have discontinued contact lens use before cross-linking and they must have undergone collagen cross-linking for keratoconus following the Dresden protocol with at least 2 years post-operative follow-up.
Patients who had previous eye surgery, underwent combined procedures (e.g. collagen cross-linking + photorefractive keratectomy, collagen cross-linking + intrastromal corneal ring segments, collagen cross-linking + refractive lenticule extraction) or had incomplete documentation due to any reason were excluded from the study.
Treatment Protocol:
The treatment procedure following Dresden protocol was conducted under sterile conditions.5 Local anesthesia was applied using proparacaine 0.5% (Alcaine®, Alcon, Belgium). Using a blunt knife, the central 7 to 9 mm of the corneal epithelium was removed. One drop of riboflavin 0.1% solution (Peschke® D, PESCHKE Trade, Switzerland) was applied to the eye every 5 minutes for 30 minutes. After this time, fluorescence was observed in the anterior chamber using slit-lamp examination. The stromal thickness at the thinnest point was then assessed by ultrasonic pachymetry. If it was at least 400 microns, UVA irradiation using the PESCHKE Trade CCL – Vario Cross-Linking (PESCHKE Trade, Switzerland) was commenced. If the stromal thickness at the thinnest point was less than 400 microns, 0.1% riboflavin and 0.9% Sodium Chloride (Euro Med, Philippines) drops were instilled every 2 minutes for 10 minutes to increase corneal thickness to greater than 400 microns before irradiation with UV light. UVA irradiation was done using two UV diodes (370 nm) with a potentiometer in series to regulate the voltage. Three 1.3-V accumulators were used as a power generator. Before each treatment, the desired irradiance of 3 mW/cm2 was controlled with a UVA meter at a 1-cm distance. The patient’s cornea was then irradiated with the UVA-light diodes at a 1-cm distance for 30 minutes using 3 mW/cm2 irradiance, which corresponds to a dose of 5.4 J/cm2. After the treatment, antibiotic and steroid drops were applied, and a bandage contact lens was fitted and kept in place until re-epithelialization occurred usually during the third to fifth day.
Primary outcome measure in this study was change in corneal curvature (Kmax). Secondary outcomes included change in corneal astigmatism, anterior and posterior elevation map changes using Pentacam® (OCULUS, Germany), and BCVA at 2 years then annually post-operatively.
Statistical Analysis:
Shapiro-Wilk test and Kolmogorov-Smirnov test with Dallal-Wikinson-Lilliefor p-value were utilized to test for normality of data sets. Parametric paired T-test and repeated measures Analysis of Variance (ANOVA) with Geisser-Greenhouse correction were utilized for comparing means in data sets with normal distribution (Kmax and BCVA). The rest of the outcome measures (corneal astigmatism measured topographically, anterior and posterior elevation map best fit sphere) were found to have normal distributions, hence, underwent parametric tests to determine significance. Non-parametric Wilcoxon matched-pairs signed rank test and Friedman test were utilized to compare means of variables with non-normal distribution. Dunn’s multiple comparisons test was utilized as a post-hoc analysis. All statistical tests were performed using GraphPad Prism 7®.
RESULTS
This study analyzed 32 patients (48 eyes) with a mean follow-up of 28.88 ± 6.23 months (Table 1). Only 4 patients (6 eyes) had follow-up up to 3 years with a mean follow-up of 42.83 ± 4.58 months. On statistical analysis, the Kmax was noted to have a non-normal distribution; hence, non-parametric tests were used to determine significance. With all eyes considered, there was a significant decrease in the corneal curvature from baseline to 2 years follow-up (55.75 ± 4.03 D vs 54.31 ± 4.19 D, p<0.0001) (Table 2). However, when only eyes with 3 years data were analyzed (n=6), there was no statistical difference between the Kmax measured at baseline and at 2- and 3- years follow-up (56.15 ± 4.78 D, 55.58 ± 5.16 D, and 55.47 ± 4.99 D, respectively, p=0.12) (Table 3).
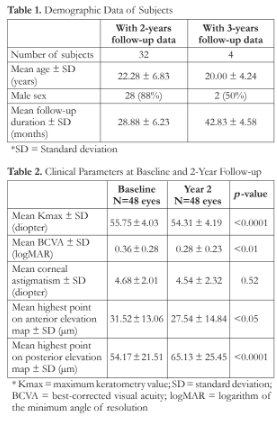

In terms of improvement in BCVA, when all eyes were analyzed, there was significant improvement in the mean BCVA from baseline to 2 years follow-up (from logMAR 0.36 ± 0.28 to logMAR 0.28 ± 0.23, p<0.01) (Table 2). When only eyes with up to 3-years follow-up were analyzed, there were improvements from baseline, but a statistically significant difference was only found between baseline and year 3 (log- MAR 0.53 ± 0.46 to logMAR 0.30 ± 0.51, p<0.05) (Table 4).

Corneal astigmatism from baseline (4.68 ± 2.01 D) to 2-year follow-up (4.54 ± 2.32 D) decreased but the difference was not statistically significant (Table 2). Also, no statistical difference was observed in corneal astigmatism measured during the pre-operative period, year 2, and year 3 (Table 3).
Analyzing the anterior surface of the cornea, there was a significant decrease between baseline and 2-year follow-up measurements (from 31.52 ± 13.06 to 27.54 ± 14.84 μm, p <0.05) (Table 2). In contrast, the posterior surface of the cornea demonstrated a significant increase of the back elevation from the baseline to 2-year follow-up (from 54.17 ± 21.51 to 65.13 ± 25.45 μm, p<0.0001). However, when only eyes with 3-year follow-up data was analyzed, there were no statistically significant changes observed for the anterior and posterior elevation maps when the baseline, 2nd and 3rd year data were compared (Table 3).
DISCUSSION
Our study showed significant flattening of the corneal curvature (Kmax) in eyes with keratoconus 2 years after cross-linking. This result is in agreement with several published studies.7-9
BCVA was also found to have significantly improved from baseline until the 3rd year post-cross-linking. This is again in line with the results from previous studies done.8,9 The improvement in visual acuity can be attributed to the significant corneal flattening, which would theoretically lead to a reduction in myopia and enable better contact lens fitting.
Our study failed to show significant changes in corneal astigmatism following collagen cross-linking. A study done by Caporossi et al. in 2010 also showed a non-significant reduction in corneal astigmatism.
A study by Steinberg et al. in 2014 utilized the highest value in the anterior and posterior elevation map to determine other possible topographic determinants in monitoring disease progression in eyes with keratoconus. Our study findings are in agreement with their results. At 2 years post-cross-linking, the anterior elevation map showed a significant decrease in the highest value while the posterior elevation map showed a significant increase in the highest value when compared to baseline measurements.
The Global Consensus on Keratoconus and Ectatic Diseases defined ectasia progression as a consistent change in at least 2 of the following parameters wherein the magnitude of the change is above the normal noise of the testing system: 1) progressive steepening of the anterior corneal surface; 2) progressive steepening of the posterior corneal surface, and 3) progressive thinning and/or increase in the rate of corneal thickness change from the periphery to the thinnest point. Our study findings demonstrated one of the criteria defining ectasia progression. However, this increase in posterior elevation may actually be due to a known decrease in the corneal thickness post – cross-linking.14 A previous study by Koller et al. in 2009 demonstrated no significant change in the posterior peak elevation post – cross-linking. While a study by Kranitz et al. in 2012 showed a significant decrease in the posterior elevation. These differences may be due to deductions in the back surface parameters being not as reliable as anterior surface parameters after corneal collagen cross-linking.14
Limitations of this study include the retrospective study design making it prone to selection bias. The low sample size obtained for those with 3-year follow–up is another limitation. It is likely due to this very low sample size that corneal curvature, corneal astigmatism, and anterior and posterior elevation map changes were found to have no significant difference when comparisons between baseline and 3-year follow-up data were made.
Overall, the results of this study support the flattening effect of collagen cross-linking on corneal curvature in eyes with keratoconus. Significant improvements in the best-corrected visual acuity and the highest value on the anterior elevation map at 2 years after cross-linking also provide support to the effectivity of corneal collagen cross-linking. The continued increase in elevation of the posterior corneal surface after collagen cross-linking may be due to a possible ongoing ectatic process or may be due to error. Future studies with a similar study design may follow-up more patients or cover a longer duration in order to further provide evidence of the efficacy of corneal collagen cross-linking for keratoconus.
REFERENCES
1. Rabinowitz YS. Keratoconus. Surv Ophthal. 1998;42(4):297- 319.
2. McGhee CN. Contemporary Treatment Paradigms in Keratoconus. Cornea. 2015;34(10S):S16 – S23.
3. Chapter 10. Corneal Dystrophies and Ectasia. Weisenthal RW, Afshari NA, Bouchard CS et al. External Disease and Cornea, Section 8. Basic and Clinical Science Course, AAO, 2017; chap. 10: pp. 378 – 384.
4. O’Brart DP. Corneal Collagen Cross Linking: A review. J Optom. 2014;7:113-124.
5. Wollensak G, Spoerl E, Seiler T. Riboflavin/Ultraviolet A induced Collagen Cross-linking for the treatment of keratoconus. Am J Ophthalmol. 2003;135(5):620-627.
6. Raiskup-Wolf F, Hoyer A, Spoerl E, Pillunat LE. Collagen Crosslinking with Riboflavin and Ultraviolet A light in Keratoconus: Long Term Results. J Cataract Refract Surg. 2008;34:796-801.
7. Raiskup-Wolf F, Theuring A, Pillunat LE, Spoerl E. Collagen Crosslinking with Riboflavin and Ultraviolet A light in Progressive Keratoconus: Ten Year Results. J Cataract Refract Surg. 2015;41(1):41-46.
8. Caporossi A, Mazzotta C, Baiocchi S, Caporossi T. Long Term Results of Riboflavin Ultraviolet A Corneal Collagen Crosslinking for Keratoconus in Italy: The Siena Eye Cross Study. Am J Ophthalmol. 2010;149(4):585-593.
9. Wittig-Silva C, Chan E, Islam FM, et al. A Randomized Controlled Trial of Corneal Collagen Cross-Linking in Progressive Keratoconus: Three-Year Results. Ophthalmology. 2014;121(4):812-821.
10. Hoyer A, Raiskup-Wolf F, Sporl E, Pillunat LE. Collagen Cross-linking with Riboflavin and UVA Light in Keratoconus: Results from Dresden. Ophthalmologe. 2009;106(2):133– 140.
11. Koller T, Mrochen M, Seiler T. Complication and failure rates after corneal crosslinking. J Cataract Refract Surg. 2009;35(8):1358–1362.
12. Sharma A, Nottage JM, Mirchia K, et al. Persistent Corneal Edema after Collagen Cross-Linking for Keratoconus. Am J Ophthalmol. 2012;154(6):922–926.
13. Sykakis E, Karim R, Evans JR, et al. Corneal collagen cross- linking for treating keratoconus (Review). Cochrane Database Syst Rev. 2015;(3):CD010621.
14. Steinberg J, Ahmadiyar R, Rost A, et al. Anterior and Posterior Corneal Changes after Crosslinking for Keratoconus. Optom Vis Sci. 2014;91(2):178-186.
15. Gomes JA, Rapuano TD, Belin MW, et al. Global Consensus on Keratoconus and Ectatic Diseases. Cornea. 2015;34(4):359–369.
16. Koller T, Iseli HP, Hafezi F, et al. Scheimpflug Imaging of Corneas After Collagen Cross-Linking. Cornea. 2009;28(5):510-515.
17. Kranitz K, Kovacs I, Mihaltz K, et al. Corneal Changes in Progressive Keratoconus After Cross-Linking Assessed by Scheimpflug Camera. J Refract Surg. 2012;28(9):645-9.

