Glycemic Control and Changes in Macular Thickness after Phacoemulsification in Diabetics
Jesa Nadine V. Protasio, MD, Romulo N. Aguilar, MD, PhD
Macular edema is a nonspecific retinal sign that may occur in association with certain systemic conditions, ocular diseases and after ocular procedures. It invariably leads to macular thickening and consequent worsening of vision. In diabetics, it occurs as a result of microvascular changes in the retina influenced by several factors including blood sugar levels.1 In postcataract surgery patients, inflammation plays an important role in its pathogenesis.2 Various studies report an increased incidence or progression of macular edema in diabetic patients following lens extraction, with a peak at 1 month post-operatively.3-6 Most cases are transient, lasting for around 6 months, although it may persist or progress in some cases.3-6 There is conflicting evidence as to the significance of glycemic control on the macula of diabetic patients following cataract surgery.3,7-9 Studies by Squirrell et al (2002), Flesner et al (2002) and Romero-Aroca et al (2006) looked at diabetic patients undergoing monocular phacoemulsification with the fellow eye serving as control. The status of the macula following cataract surgery was assessed using various methods (slit lamp biomicroscopy and fluorescein angiography) and results revealed no significant association with HbA1c.7-9 However, a study by Hayashi et al in 2009 yielded conflicting results. This study compared eyes with and without diabetic retinopathy who underwent phacoemulsification. The post-operative status of the macula was assessed using Stratus OCT, and this study concluded that HbA1c at the time of operation in eyes with diabetic retinopathy was significantly greater than in those eyes without, and correlated significantly with foveal thickness. Another conclusion of Hayashi’s study is that HbA1c may be a significant predictor for progression of macular edema.3 These studies utilized glycosylated hemoglobin or HbA1c as an indicator of systemic glycemic control. HbA1c has been accepted as the gold standard with regards to assessment and monitoring of glycemic control in diabetics. It reflects the average blood sugar level in the past 3 months. The target for attainment of glycemic control has been set at HbA1c ≤6.5%. Since the status of the macula is the primary predictor of visual outcome following cataract surgery3 , it is of value to look at modifiable factors such as glycemic control that may reduce the incidence of this postoperative complication.
The general objectives of this study are to measure the mean central macular thickness (CMT) in diabetics who undergo monocular phacoemulsification, and to determine the association between perioperative glycemic control and mean CMT after phacoemulsification in diabetics. Specifically, this study aims to determine baseline CMT of both eyes using the Cirrus 5000 OCT, to measure the CMT of both eyes 1 month after phacoemulsification of the study eye, to assess glycemic control during the study period by measuring HbA1c levels 1 month after phaco emulsification, to correlate mean CMT in the study and control eyes with HbA1c levels, and to correlate changes in CMT with BCVA in the study and control eyes using an Early Treatment Diabetic Retinopathy Study (ETDRS) chart. If such an association is established, this will be of significance to cataract surgeons dealing with diabetic patients in terms of immediate post-operative visual prognosis. A surgeon may then opt to wait until good glycemic control is achieved prior to operating on a diabetic patient, despite clearance for surgery by an internist or endocrinologist, in order to achieve optimal results. In the same manner, a diabetic patient, who opts to undergo phacoemulsification in spite of suboptimal glycemic control, may also be advised regarding realistic immediate postoperative expectations.
METHODOLOGY
This was a prospective cohort study conducted at the University of the Philippines Department of Ophthalmology and Visual Sciences (UP-DOVS) – Sentro Oftalmologico Jose Rizal (SOJR) of the Philippine General Hospital (PGH) from March to August 2015. Participants included in the study were screened using set inclusion and exclusion criteria. The inclusion criteria included diabetic patients (Type 1 or Type 2) with or without documented retinopathy, with or without pre-existing macular edema, seen at the General, Cataract or Medical Retina Clinics of SOJR who were scheduled for phacoemulsification. They must have had vision equal to or less than 20/30 due to cataract in at least one eye. Also, they must have been medically cleared to undergo cataract surgery, willing to have blood drawn, willing to undergo optical coherence tomography, willing to participate in the study, and with at least verbal consent from the cataract surgeon to include their patient in the study. Patients were excluded from the study if they had undergone previous ocular surgery, had severe media opacity of either eye resulting in inaccurate OCT measurements, had suspected or documented macular pathology not due to diabetes, and if they underwent complicated phacoemulsification (specifically, had a posterior capsule break or tear, vitreous loss, dropped lens material or intraocular lens, tear of Descemet’s membrane, expulsive suprachoroidal hemorrhage, procedure lasting more than 1 hour from cutting time, and surgeon-assessed difficulties in the procedure). Thirty-five patients were referred for this study. Eighteen were referrals from the Cataract Clinic while 17 were referred from the Medical Retina Clinic of the Philippine General Hospital Sentro Oftalmologico Jose Rizal (PGH-SOJR). Patients who were eligible for the study based on the aforementioned inclusion and exclusion criteria were invited to participate and asked to sign a written consent form. Nine patients were excluded; eight because of cataracts that were too dense such that the OCT did not give a reliable signal, while one was excluded because of complicated surgery. Baseline BCVA of both eyes was determined by the principal investigator using an ETDRS chart at a period of up to 8 weeks before scheduled phacoemulsification of the study eye. On the same day, the patient underwent OCT of the macula of both eyes using the Cirrus 5000 OCT, at the second floor Eye Instrument Clinic (EIC) of PGH SOJR. Cataract surgery was performed as scheduled by the senior ophthalmology resident assigned to the patient. The patient also followedup with his/her surgeon as deemed necessary by the latter. For purposes of this study, BCVA of both eyes was recorded using an ETDRS chart at 1 month postoperatively. At this time, patient also underwent OCT of both eyes, as well as blood extraction for HbA1c assessment. Eleven patients dropped out from the study; one patient underwent phacoemulsification of the other eye before the 1 month mark as per patient’s request, while 10 patients were lost to follow-up. This left a final sample size of 15, data from which was statistically analyzed. Primary outcomes for this study were CMT of the study and control eye at baseline and at 1 month post-phacoemulsification measured using the Cirrus 5000 OCT, and perioperative glycemic control determined by HbA1c levels taken 1 month after phacoemulsification. The secondary outcome was BCVA in logmar units using the ETDRS chart obtained at baseline and at 1 month after phacoemulsification. Statistical Analysis Paired T-test was done to compare the mean CMT and BCVA at baseline and at 1 month after phacoemulsification within and between the cohorts of study and control eyes. Unpaired T-test was used to compare the mean change in CMT in the study and control eyes (different samples: study eyes vs control eyes). Paired T-test was used to compare changes in CMT per patient against HbA1c. Simple correlation between HbA1c and CMT, as well as CMT and BCVA, was done using Pearson’s R.
RESULTS
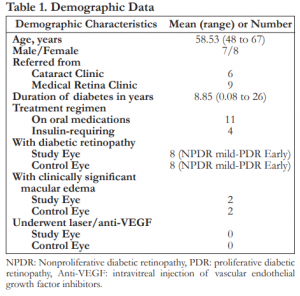
A total of 15 patients were included in the study. Mean age in years was 58.53 with ages ranging from 48 to 67 years old. There were seven males and eight females. Six were referred from the Cataract Clinic while nine were referred from the Medical Retina Clinic. Mean duration of diabetes in years is 8.85. Eleven were maintained on oral medications while four received insulin with or without oral medications. Eight patients had diabetic retinopathy of both eyes, two of which had clinically significant macular edema. None of the patients have undergone photocoagulation or anti-VEGF injections (Table 1).
Ten right eyes and five left eyes were included in the study group. Patients were operated on by five senior residents. Baseline CMT was taken at a mean of 9.27 days before phacoemulsification while post-operative CMT was taken at a mean of 28.8 days after surgery. Intraocular lenses implanted were Alcon Acrysof in seven, Rohto in seven, and Ocuflex in one. All 15 study eyes were treated with prednisolone and moxifloxacin drops, with three patients having to instill a third medication – sodium chloride, as deemed necessary by their surgeon (Table 2).

Baseline and post-op CMT of both the study and control eyes were not statistically different with p=0.8771 and p=0.1562 respectively. In the cohort of study eyes, we noted a greater mean CMT postoperatively (p=0.004). In the cohort of control eyes, the CMT after 1 month did not significantly differ from baseline CMT (p=0.442) (Table 3 and Figure 1).


Thirteen of fifteen study eyes (86.67%) had an increase in CMT compared to baseline but in twelve of these patients (80%), CMT values were still within normal range. Only in one study eye (6.67%) did the thickening progress to macular edema. The patient with the greatest increase in CMT (67 μm) had a pre-operative diagnosis of non-proliferative diabetic retinopathy (NPDR), moderate. A patient diagnosed with proliferative diabetic retinopathy (PDR) did not have an increase in CMT after surgery. The mean change in CMT in the cohort of study eyes was 16.7 ± 18.5 μm, while it was 1.8 ± 8.8 μm in the group of control eyes. The difference in these mean changes in CMT was statistically significant with a p-value of 0.011 (Table 4 and Figure 2).

Comparing the changes in CMT versus HbA1c per patient, there is a weak negative correlation that is not significant at p<0.05 for both study and control eyes (r=-0.1238 and r=-0.331, respectively)
(Figures 3 and 4).

Mean HbA1c was 8.19%, ranging from 5.7% to 12.2%. Only one patient had a normal HbA1c at 5.7%. No positive correlation was demonstrated between HbA1c and mean CMT in the group of study eyes, r=-0.312 (Figure 5). The patient with the most thickening at 301.5 μm only had an HbA1c of 7.4%, while the study eye of the patient with the highest HbA1c (12.2%) only had a mean CMT of 214.

Baseline BCVA was statistically different in the eyes that were to undergo an operation from those eyes that served as control (p=0.023). Mean postoperative BCVA was significantly different from baseline in the cohort of study eyes (p=0.002) (Table 5).


DISCUSSION
According to current literature, development or progression of macular edema in 20 to 50% of eyes that undergo phacoemulsification peak at 4 to 6 weeks.3,7 This is contrary to what we observed in our data where only one eye (6.67%) progressed to macular edema. In 12 other eyes (80%), CMT increased but was still within normal range. Reasons as to why this trend was not observed in our study include a limited sample (target sample size was 50) and the timing of OCT. Although literature describes a peak increase in macular thickness at 4 to 6 weeks after surgery, our study may have missed out on possible delayed-onset macular thickening. We note that these studies had long-term follow-ups for up to 12 months which gave them ample time to catch trends in CMT changes. Available literature report an increase in foveal thickness greater in eyes with diabetic retinopathy than in those without. This is attributed to the release of cytokines from the blood-ocular barrier after surgery, coupled with the increased breakdown of the blood-ocular barrier in diabetic eyes.3,7 Again, this is contrary to what we observed in our data where the patient with the most increase in CMT (67 μm) was only given a pre-operative diagnosis of NPDRmoderate while the patient with proliferative diabetic retinopathy did not have an increase in CMT after surgery. The same limitations of a small sample size and timing of OCT may explain these observations. The cohort of study eyes had a greater change in CMT (16.7 ± 18.5 μm) compared to the cohort of control eyes (1.8 ± 8.8 μm) which was statistically significant at p=0.011. We observe the role of surgery in the cohort of study eyes because any change in the fellow eye would be assumed to be brought about by the disease itself. Seeing how the data we gathered compares to recent studies (Table 6), we note similar baseline and post-operative CMT in diabetic eyes who undergo phacoemulsification when we compared our results with those gathered by the Pan American Collaborative Retina Study Group (PACORES).10 Looking at the amount of CMT increase in diabetic eyes that underwent uncomplicated phacoemulsification, we noted similar changes when we compared our results with that gathered by the PACORES group and in the no diabetic retinopathy group in the study conducted by Kim et al. (2007).11

No positive correlation was established between mean CMT in the study and control eyes. Our data showed that an increased HbA1c did not necessarily parallel an increased mean CMT. This echoes some studies in available literature such as those of Squirrell et al (2002), Flesner et al (2002), and Romero-Aroca et al (2006) that said that the status of the macula had no significant association with HbA1c.7-9 This contradicts what Hayashi et al (2009) said that HbA1c at the time of operation correlated significantly with foveal thickness, at least in their cohort of eyes with diabetic retinopathy, and that HbA1c may be a predictor for progression of macular edema.3 The absence or lack of positive correlation between HbA1c and mean CMT suggest the presence of confounding variables. These include the number and expertise of the surgeons (five beginning cataract surgeons with different phacoemulsification techniques); the density of the cataract to begin with and the resulting ultrasound power and duration of phacoemulsification required to remove the cataract; the type of intraocular lens placed; the frequency, duration and compliance to medications; and the fact that our sample population was skewed towards those with abnormal HbA1c. An incidental finding of value, however, is the fact that most of these diabetic patients who were cleared for cataract surgery actually had deranged HbA1c levels reflecting poor glycemic control. While we failed to demonstrate a positive correlation between HbA1c and change in CMT after phacoemulsification in this study, we still think it is of value to look at a possible correlation between the two as they have implications for the future care of diabetic patients with cataracts. With regards to our secondary outcome, BCVA, we saw that baseline BCVA was statistically different in the eyes that were to undergo surgery from those that served as control. This is expected because we would assume worse baseline BCVAs in the study eyes due to cataract. Mean post-op BCVA was statistically different from baseline in the cohort of study eyes which can be explained by the surgery itself. No significant conclusions can be derived from this. A weak positive correlation between BCVA and CMT was demonstrated. This echoes what is said in available literature, that is, worsening of macular edema after cataract surgery decreases visual acuity only in some cases.7-9 We acknowledge the limitations of this study. Although there is a large pool of patients to choose from (diabetics with cataracts), we were not able to reach the target sample size. This was due to a lot of factors including cataracts that were too dense (affecting the OCT signal and thus the accuracy of the CMT measurement), unavoidable complications during surgeries, patients who were in a hurry to have their other eye operated on or those who had immediate indications for removal of cataract in their other eye (such as panretinal photocoagulation or fluorescein angiography), and those who did not want to follow-up. Also, patients ideally could have been grouped into those with controlled HbA1c and those with uncontrolled HbA1c, and a comparison between their mean changes in CMT could have been made. Since determining HbA1c pre-operatively is not the standard of care (nor is it required for pre-operative clearance in our institution), we had to tailor this study in order to satisfy requirements of the ethical board (who had issues regarding the invasiveness of the procedure, as well as the implied increase in cost of the study). In the process, we did not get our ideal cohort of patients. Recommendations for further studies include the construction of a well-structured streamlined study design, meeting the target sample size, and separating the participants into two cohorts based on controlled and uncontrolled HbA1c. A longer post-operative period may also be helpful so as not to miss out on those with possible delayed CMT changes and so that possible trends can be observed.
CONCLUSION
Change in CMT in diabetic eyes that undergo phacoemulsification is significant. This was demonstrated when we compared the mean change in CMT in the eyes that underwent surgery (study eyes) and compared it to the cohort of control eyes. The significant difference between the two cohorts is suggestive of the influence of the surgery itself. While it is common for CMT to increase in diabetics who undergo phacoemulsification, this may not necessarily result in macular edema, nor does it necessarily parallel severity of diabetic retinopathy. We were not able to demonstrate a correlation between CMT and HbA1c levels among diabetics whose eyes underwent uncomplicated phacoemulsification, nor were we able to demonstrate a correlation between CMT and BCVA.
ETHICAL CONSIDERATIONS
This study was conducted in full conformance proper. All participants underwent cataract surgery, which if uneventful, improved vision. As with any surgery, unforeseen complications may arise which can compromise visual outcome. These risks were explained to the patient by the investigator and the surgeon. Macular thickness measured using the Cirrus 5000 OCT was taken twice: pre-operatively and postoperatively at 1 month. HbA1c was determined at 1 month post-phacoemulsification which was reflective of glycemic control for the past 3 months, inclusive of the entire study period. There were no foreseen risks or complications in the conduction of these tests. If a participant’s vision or medical condition became compromised at any point during the study, or if the aforementioned diagnostic tests revealed results that necessitated intervention, this was not withheld, and the participants were withdrawn from the study and treated as a dropout. Participants were also aware that they could withdraw from the study at any time. There was no conflict of interest in the conduct of this study. The principal investigator, together with the UPDOVS SOJR shouldered all expenses that arose from this research endeavor. with principles of the “Declaration of Helsinki”, Good Clinical Practice (GCP) and within the laws and regulations of the University and the country. This protocol was submitted for ethical evaluation to the University of the Philippines Manila Research Ethics Board (UPMREB) and was conducted only when approval was granted. The participants’ interests were held with utmost importance. All identifying patient information was kept confidential. Informed consent from the participant was obtained prior to the study proper. All participants underwent cataract surgery, which if uneventful, improved vision. As with any surgery, unforeseen complications may arise which can compromise visual outcome. These risks were explained to the patient by the investigator and the surgeon. Macular thickness measured using the Cirrus 5000 OCT was taken twice: pre-operatively and postoperatively at 1 month. HbA1c was determined at 1 month post-phacoemulsification which was reflective of glycemic control for the past 3 months, inclusive of the entire study period. There were no foreseen risks or complications in the conduction of these tests. If a participant’s vision or medical condition became compromised at any point during the study, or if the aforementioned diagnostic tests revealed results that necessitated intervention, this was not withheld, and the participants were withdrawn from the study and treated as a dropout. Participants were also aware that they could withdraw from the study at any time. There was no conflict of interest in the conduct of this study. The principal investigator, together with the UPDOVS SOJR shouldered all expenses that arose from this research endeavor.
REFERENCES
1. Schubert H, Atebara N, Kaiser R, Martidis A, McCannel C, Zacks D, Dhindsa H. Retinal vascular disease: Diabetic Retinopathy. American Academy of Ophthalmology. San Francisco: American Academy of Ophthalmology, 2012; Section 12 Retina and Vitreous: 89-112.
2. Flach AJ. The incidence, pathogenesis and treatment of cystoid macular edema following cataract surgery. Trans Am Ophthalmol Soc 1998;96:557-634.
3. Hayashi K, Igarashi C, Hirata A, et al. Changes in diabetic macular oedema after phacoemulsification surgery. Eye (Lond) 2009;23;389-96.
4. Kwon SI, Hwang DJ, Seo JY, et al. Evaluation of changes of macular thickness in diabetic retinopathy after cataract surgery. Korean J Ophthalmol 2011;25:238-42.
5. Eriksson U, Alm A, Bjärnhall G, et al. Macular edema and visual outcome following cataract surgery in patients with diabetic retinopathy and controls. Graefes Arch Clin Exp Ophthalmol 2011;249:349-59.
6. Kim SJ, Equi R, Bressler NM. Analysis of macular edema after cataract surgery in patients with diabetes using optical coherence tomography. Ophthalmology 2007;114:881-9.
7. Squirrell D, Bhola R, Bush J, et al. A prospective, case controlled study of the natural history of diabetic retinopathy and maculopathy after uncomplicated phacoemulsification cataract surgery in patients with type 2 diabetes. Br J Ophthalmol 2002;86:565-71.
8. Flesner P, Sander B, Henning V, et al. Cataract surgery on diabetic patients. A prospective evaluation of risk factors and complications. Acta Ophthalmol Scand 2002;80:19-24.
9. Romero-Aroca P, Fernández-Ballart J, Almena-Garcia M, et al. Nonproliferative diabetic retinopathy and macular edema progression after phacoemulsification: prospective study. J Cataract Refract Surg 2006;32:1438-44.
10. Gallego-Pinazo R, Dolz-Marco R, Berrocal M, et al. Outcomes of cataract surgery in diabetic patients: results of the Pan American Collaborative Retina Study Group. Arq Bras Oftalmol 2014;77:355-9.
11. Kim SJ, Equi R, Bressler NM. Analysis of macular edema after cataract surgery in patients with diabetes using optical coherence tomography. Ophthalmology 2007;114:881-9.
12. Hannouche RZ, Avila MP. Retinal thickness measurement and evaluation of natural history of the diabetic macular edema through optical coherence tomography. Arq Bras Oftalmol 2009;72:433-8.
13. Kang SW, Park CY, Ham DI. The correlation between
fluorescein angiographic and optical coherence tomographic features in clinically significant diabetic macular edema. Am J Ophthalmol 2004;137:313-22.
14. Knez N, Šiško K, Pahor D. Influence of cataract surgery on macular thickness – a 3-month follow-up. J Int Med Res 2011;39:1113-21.
15. Yeung L, Sun CC, Ku WC, et al. Associations between chronic glycosylated haemoglobin (HbA1c) level and macular volume in diabetes patients without macular oedema. Acta Ophthalmol 2010;88:753-8.
16. Kiernan DF, Hariprasad SM. Normative databases in SDOCT: a status report. Retinal Physician 2010;1-18.
17. Kilpatrick ES. HbA1c measurement. J Clin Pathol 2004;57:344-5.
18. Cobin R, Davidson J, Ganda O, Garber A, Hellman R, Jellinger P, Levetan C, Palumbo P, Rodbard H. American College of Endocrinology consensus statement on guidelines for glycemic control. Endocr Pract 2002;8(Suppl 1):5-11.

