An Economic Analysis of Selective Laser Trabeculoplasty versus Topical Prostaglandin Analogues as Initial therapy for Primary Open-Angle Glaucoma in a Tertiary Government Hospital in the Philippines
James Michael D. Jacomina, MD, John Mark S. de Leon, MD,
Jose Maria D. Martinez, MD-MBA
Department of Health (DOH) Eye Center, East Avenue Medical Center, Quezon City
Correspondence: James Michael D. Jacomina, MD
Email Address: jamesjacomina@gmail.com
Clinic Address: DOH Eye Center, East Avenue Medical Center, Diliman, Quezon
City Clinic Phone Number: +639178552948
Disclosures: The authors have no financial interests or conflicts of interest to report.
Glaucoma is the second leading cause of irreversible blindness in the world. Primary open angle glaucoma (POAG) is the most prevalent glaucoma subtype with world-wide prevalence of 52.68 million in 2020.1 Globally, 90% of overall blindness is concentrated in the developing countries.2 In the Philippines, glaucoma is the third leading cause of bilateral blindness and the leading cause of permanent blindness.3
Topical hypotensive medications is the standard first-line treatment for POAG while selective laser trabeculoplasty (SLT) is emerging as an alternative treatment that can delay, prevent, or reduce the need for glaucoma eye drops.4 Prostaglandin analogues (PGAs) are frequently used glaucoma eye drops and are considered to be the most potent among the different drug classes in reducing intraocular pressure (IOP).5 SLT is a 15-minute clinic-based laser procedure that targets and improves outflow through the trabecular meshwork resulting in 25% IOP reduction at 89% efficiency.6
Initially, SLT was available only in private institutions mostly located in the urban areas in the Philippine National Capital Region (NCR). The Department of Health Eye Center (DOHEC) of the East Avenue Medical Center (EAMC) acquired the SLT in 2015. A local SLT economic study highlighted the difference between the one-time initial SLT costs and the recurrent costs of conventional topical glaucoma eye drop medications.7
Several studies have compared the cost of treatment of PGAs and SLT within a variety of health systems. In the United States, Cantor et al. found that the cost of treatment over 5 years was cheapest for SLT compared to PGAs or surgery.8 Stein et al. reported similar findings wherein the cost per Quality Adjusted Life Years (QALY) gained was $14,179 for PGAs, and $16,824 for SLT.9 In Canada, the cost of SLT was lower than multiple glaucoma drug therapy.10 Similarly, Gazzard et al. reported that in the United Kingdom, topical glaucoma treatment costs more than twice compared to SLT over three years.4 Meanwhile, Wittenborn et al. established that topical glaucoma medications had higher cost per disability adjusted life years (DALY) compared to SLT in Barbados and Ghana.11 In the Philippines, Martinez found that the cost-effectiveness projected over one year in private settings was Php 2,901.65/mmHg IOP reduction for PGAs vs. Php 4,964.44/mmHg IOP reduction for SLT. In this case, PGAs were more cost effective than SLT over a year in a private setting; however, once senior citizen discounts or Philippine Health Insurance Corporation (PHIC) benefits were applied, the cost-effectiveness improved for both treatment modalities at Php 2321.32/mmHg IOP reduction for PGAs and Php 2047.74/mmHg IOP reduction for SLT. In this case, SLT was slightly more cost-effective than PGAs.7
In the Philippines, the cost of outpatient glaucoma eye drops is paid primarily out-of-pocket, while full coverage is given by the government-run PHIC for SLT as well as for the different glaucoma surgeries on a case-rate basis. In the setting of a government training institution, there is no additional out-of-pocket cost for professional fees since the treatment of patients is done pro bono by residents-in-training. In addition, since glaucoma is a long-term progressive disease, we would like to know the economic benefit of PGAs versus SLT for an extended period of time.
This study aimed to determine costeffectiveness of SLT versus topical PGAs as initial treatment for patients diagnosed with POAG.
METHODS
This study was an economic analysis using actual, current treatment costs of PGA therapy and SLT applied to theoretical, literature derived clinical efficacy data projected for a period of 19 years. Adult patients of the DOHEC-EAMC diagnosed with POAG who were on any topical antiglaucoma medication and had undergone SLT or glaucoma drainage surgery were surveyed from March to April 2022. In order to be included in the study, they must comprehend either Filipino or English. Patients who had concomitant ocular diseases (retinal, corneal pathology) were excluded from the study. Our study adhered to the basic principles of the Declaration of Helsinki and conformed with the guidelines set forth by the International Council for Harmonization Good Clinical Practice (ICH-GCP). The EAMC Institutional Ethics Review Board approved this study.
Purposive sampling was utilized in this study. Sample size calculation was carried out with a confidence level of 95% and a margin of error of 5%. Estimated sample size was 29 participants.
A face-to-face survey which included demographics and household income and spending was conducted by the principal investigator. Indirect medical costs were calculated by multiplying the cost of transportation to the eye center (jeep, bus, train, or car) and the glaucoma diagnostic tests (visual fields and/or optical coherence tomography) with its frequency per year which was then added to the annual cost of SLT/PGAs to get the total annual cost of each intervention per person.
The PGA glaucoma eyedrop included in this study were Lumigan® (Bimatoprost, Allergan, Inc. Irvine, California, USA), Xalatan® (Latanoprost, Pfizer, Inc., New York City, New York, USA), Travatan® (Travoprost, Alcon Laboratories, Inc. Fort Worth, Texas, USA) and Taflotan® (Tafluprost, Santen Pharmaceutical, Co., Ltd, Higashiyodogawaku, Japan). The retail prices of these 4 eye drops were obtained from a single pharmacy during the period of March to April 2022. The average price per bottle was multiplied by the bottles consumed per person per year to calculate the annual cost of the bottles per person.12 Treatment cost of SLT (SLT Deux®, Lightmed, San Clemente, California, USA) was obtained from the price list of the DOHEC and its equivalent PHIC coverage.
Cost-effectiveness was calculated by dividing the total annual cost per person by the average IOP reduction based on literature for each treatment modality (cost in Php per mmHg IOP reduction). Cost-utility was calculated given the following scenarios: average age at glaucoma diagnosis of 60 years13 and the life expectancy of Filipinos at 79 years which equates to additional 19 years after glaucoma diagnosis.14
First scenario is the diagnosed but untreated theoretical cohort which is a patient who is diagnosed with POAG at 60 years old and goes untreated. This patient will live with a utility value of 0.97 upon diagnosis and will develop very poor vision of counting fingers (CF) at around age 71 years.15 This patient will then live an additional 8 years with a utility of 0.52 until his or her death.
Second scenario is the treated theoretical cohort which is a patient who is diagnosed with POAG at 60 years and treated with either SLT or PGAs with good IOP control and minimal side effects with a utility value of 0.95.16 There may be additional SLT applications as well as adjunctive glaucoma eye drops but the quality of vision and life would have been preserved until death at 79 years.
We assumed the utility values for SLT and PGAs were similar. The QALY was calculated. The total direct costs were projected at 19 years taking into account costs and outcomes at different times.
Cost per QALY was calculated using the KeelerCretin paradox formula including the projected improvement in cost of health care at 3% per year.
Present Value of Cost
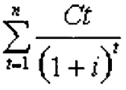
Note that Ct represents the cost incurred in year t, and that i is the discount rate
The respective values were then calculated over a time horizon of 19 years to get the cost utility ratios and direct medical costs.
RESULTS
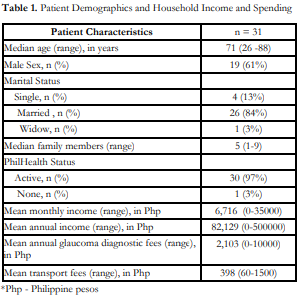
This study surveyed 31 patients with POAG. The patient demographics are listed in Table 1.
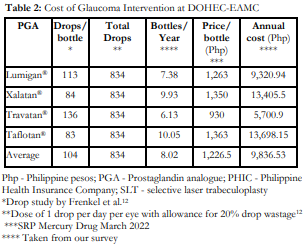
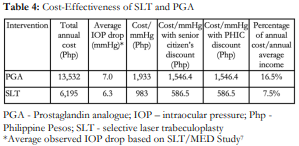
The average annual cost of PGAs is Php 9,836.53 (Table 2). The average annual cost of SLT at our institution is Php 2,500. Each of the treatment costs above was added to the indirect medical cost of Php 3,695 (Table 3) to compute the total annual cost for each intervention. SLT had a total annual cost of Php 6,195 compared to Php 13,532 for PGAs.
Cost-effectiveness was Php 1,933/mm Hg IOP reduction for PGA and Php 983/mm Hg IOP reduction for SLT. When PHIC coverage was accounted for, cost-effectiveness was Php 1,546.4/mmHg IOP reduction for PGA and Php 586.5/mmHg IOP reduction for SLT (Table 3).

The cost-utility analysis (Table 5) shows 4.3 QALY gained from initiating either SLT or PGA with a cost utility ratio of Php 27,373/QALY for SLT compared to Php 59,793/QALY gained for PGAs. If a patient had an active PHIC membership, the cost per QALY gained would be Php 16,327 for SLT and Php 47,831 for PGAs.
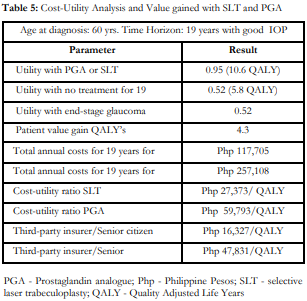
The total annual cost projected at 19 years for treating POAG with PGAs is Php 257,108, and with SLT is Php 117,705 (Table 5). If a third-party insurer is involved, the total annual cost would be Php 205,671 with PGAs and Php 70,204 with SLT.
DISCUSSION
The Philippine healthcare system can be classified as a combination of two health care models, the Bismarck model (social health insurance with premium paying by employers/members) represented locally by the PHIC, and the out-of-pocket model. Some countries with exclusive Bismarck models are Germany, Belgium, Japan, and Switzerland while the Beveridge model (with national health service that funds free healthcare) is seen in the United Kingdom. A combination of both Bismarck and Beveridge models is seen in Canada, Taiwan and South Korea wherein the government provides payment for the insurance program driven by private providers.
Among glaucoma patients, the economic analysis would differ in various countries depending on factors such as cost of medications, cost of laser, and health insurance coverage. Considering the healthcare system in resource-challenged settings such as in the Philippines, it would be very beneficial to identify the most cost-effective glaucoma treatment modality.
Our hospital-based cohort survey of 31 glaucoma patients showed that the average household monthly income was only Php 6,716. Thus, the annual household income of our cohort was Php 82,129 compared with Php 313,348 obtained from a population-based 2018 Philippine Statistics Authority survey.15 Our cohort thus seemed to represent the poorest of the poor. Even though 97% of the patients included in our study had active PHIC membership, the cost of PGAs would have to be paid out-of-pocket since PHIC does not cover these medications. In our patient base, the annual cost of glaucoma treatment represents 7.5-16.5% of their household income (Table 4). In this situation, it is easy to understand that subsidized one-time effective treatment costs are more favorable than non-subsidized recurring treatment costs, assuming these were as effective. Looking at the projected annual costs for 19 years (Table 5), treatment costs with PGAs would almost be double that of SLT. Given a similar number of QALY gained between the two treatment modalities, patients pay less with SLT at Php 27,373/QALY gained compared with Php 59,793/QALY gained for PGAs.
The local 2015 thesis of Martinez conducted in private hospitals showed that PGAs were more costeffective than SLT in one year; however, if PHIC coverage and senior citizen discount were applied, SLT was slightly more cost-effective.7 Our 2022 study showed that in a government eye center setting projected over 19 years, SLT was more cost-effective by an even larger margin given the big difference in facility cost.
Similarly, studies in developing countries such as Ghana showed that SLT had a cost per DALY of $1,771 compared to topical medications that had a cost per DALY of $6,896, and likewise in Barbados SLT had a cost per DALY of $1,528 and topical medications had a cost per DALY of $7,728.11
Cost-effective health care attempting to reduce the disease burden of visual loss is the priority of all nations but notably in the developing world since this may pose an economic burden to society. Even if the cost of SLT is covered with active PHIC membership, the burden of payment still falls on the shoulders of the payors and their active PHIC contributions. PHIC membership is automatic for senior citizens (≥ 60 years of age) in the Philippines; however if the patient is below 60 years of age, he or she will not be able to avail of the benefit of SLT unless he or she had applied for membership earlier with proof of PHIC premium contributions. Nonetheless, the cost utility ratios seem more favorable for SLT than PGAs, either with or without this third-party coverage. Taking into consideration the socioeconomic status of our patients and the costs of both procedures in POAG treatment, SLT can be offered as initial therapy for these patients with or without active PHIC membership status due to the long-term savings that can be accrued.
This economic analysis shows that SLT is the more cost-effective treatment option in newly diagnosed POAG in a low-resource, public hospital setting. SLT can be offered as initial therapy for POAG to decrease the economic burden for patients as well as the healthcare system.
REFERENCES
- Zhang N, Wang J, Li Y, Jiang B. Prevalence of primary open-angle glaucoma in the last 20 years: A meta-analysis and systematic review. Sci Rep. 2021 Jul 2;11(1):13762.
- Quigley HA. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol. 2006;90(3):262–7.
- Cubillan LDP, Olivar-Santos EO. Third National Survey on Blindness. Phillip J Ophthalmol. 2005;30(3):100–14. 4.
- Gazzard G, Konstantakopoulou E, Garway-Heath D, et al. Selective Laser trabeculoplasty versus drops for newly diagnosed ocular hypertension and glaucoma: The LIGHT RCT. Health Technol Assess. 2019;23(31):1–102.
- Stewart WC, Konstas AGP, Nelson LA, et al. Meta-analysis of 24-hour intraocular pressure studies evaluating the efficacy of glaucoma medicines. Ophthalmology. 2008;115(7):1117-1122.
- Katz LJ, Steinmann WC, Kabir A, et al. Selective Laser trabeculoplasty versus medical therapy as initial treatment of glaucoma. J Glaucoma. 2012;21(7):460–8.
- Martinez JM. Economic Analysis of Selective Laser Trabeculoplasty versus Conventional Medical Therapy in the Initial Treatment of Primary Open Angle Glaucoma. Health Economics for Managers, MBA. Health H01 Ateneo Graduate School of Business 2015:1-9.
- Cantor LB, Katz LJ, Cheng JW, et al. Economic evaluation of medication, laser trabeculoplasty and filtering surgeries in treating patients with glaucoma in the US. Curr Med Res Opin. 2008;24(10):2905-18.
- Stein JD, Kim DD, Peck WW, et al. Cost-effectiveness of medications compared with laser trabeculoplasty in patients with newly diagnosed open-angle glaucoma. Arch Ophthalmol. 2012;130(4):497-505.
- Lee R, Hutnik CML. Projected cost comparison of selective laser trabeculoplasty versus glaucoma medication in the Ontario Health Insurance Plan. Can J Ophthalmol. 2006;41(4):449–56.
- Wittenborn JS, Rein DB. Cost-effectiveness of glaucoma interventions in Barbados and Ghana. Optom Vis Sci. 2011;88(1):155-63.
- Frenkel REP, Frenkel M, Toler A. Pharmacoeconomic analysis of prostaglandin and prostamide therapy for patients with glaucoma or ocular hypertension. BMC Ophthalmol. 2007;7:16.
- Li Yim J, Ng J, Ting N. Different strategies and costeffectiveness in the treatment of Primary Open-Angle Glaucoma. Clinicoecon Outcomes Res. 2014;6:523-30.
- Philippine Statistics Authority. 2018 Family Income and Expenditure Survey. National and Regional Estimates. Volume I. https://psa.gov.ph/sites/default/files/FIES%202018% 20Final%20Report.pdf (accessed March 15, 2023).
- Brown GC, Stein JD, Brown MM, Wilson RP, Spaeth GL. Landmark Studies. Glaucoma Therapy Delivers Great Patient and Financial value. Glaucoma Today. 2014 July/August:13–8.
- Garg A, Gazzard G. Selective Laser Trabeculoplasty: Past, present, and future. Eye. 2018;32(5):863–76.

